Sorghastrum secundum
| Sorghastrum secundum | |
|---|---|

| |
| Photo by Kevin Robertson | |
| Scientific classification | |
| Kingdom: | Plantae |
| Division: | Magnoliophyta - Flowering plants |
| Class: | Liliopsida - Moncots |
| Order: | Poales |
| Family: | Poaceae |
| Genus: | Sorghastrum |
| Species: | S. secundum |
| Binomial name | |
| Sorghastrum secundum (Elliott) Nash | |
| |
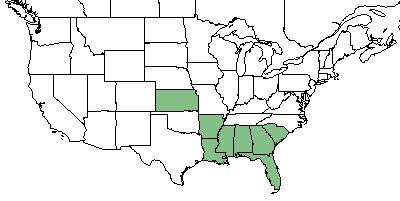
| Natural range of Sorghastrum secundum from USDA NRCS Plants Database. | |
Common Name(s): lopsided indiangrass[1][2]
Contents
Taxonomic Notes
Synonym(s): Andropogon secundus[2]
Description
Sorghastrum secundum is a monoecious perennial graminoid[2] that grows to 3-6 feet (0.91-1.83 m).[3] It is a tufted grass with short rhizomes.[4] Culms are erect to ascending and blades are up to 8.1 mm (0.32 in) wide and 42 cm (1.38 ft) long. Straight inflorescence with spikelets positioned all on one side helps distinguish this species.[5]
Sorghastrum secundum does not have specialized underground storage units apart from its rhizomes.[6] Diaz-Toribio and Putz (2021) recorded this species to have an non-structural carbohydrate concentration of 22 mg/g (ranking 94 out of 100 species studied).[6]
According to Diaz-Torbio and Putz (2021), Sorghastrum secundum has rhizomes with a below-ground to above-ground biomass ratio of 1.04 and nonstructural carbohydrate concentration of 22 mg g-1.[7]
Distribution
Sporobolus floridanus is endemic to an area from southern South Carolina to peninsular Florida and west through Alabama, but the majority is found in Florida.[8]
Ecology
Habitat
S. secundum has been found in pine-oak-hickory woods, longleaf pine slopes, longleaf pine savanna, pineland grasses, live oak-sand pine woods, wet pine flatwoods, and longleaf pine-scrub oak ridges.[9] It is also found in disturbed areas including burned longleaf pinelands and the disturbed sands of hammock clearings.[9]
Associated species: Serenoa repens, Balduina angustifolia, Amphicarpum muhlenbergianum, and Sisyrinchium solstitiale.[9][10]
This species is found on sandhill and other upland pine communities[1] and thinly wooded areas.[5] In peninsula Florida xeric sandhills, S. secundum was found in 100% of the plots with a mean cover of 0.0299 m2. Panhandle xeric sandhills contained S. secundum in 87% of plots and had a mean coverage of 0.0191 m2. Frequency and mean cover in northern Florida longleaf ("clayhill") woodlands was 100% and 0.04 m2 and 94% and 0.0273 m2 in northern Florida subxeric sandhills, respectively.[11]
S. secundum has reduced its occurrence in response to agricultural-based soil disturbance in historically longleaf communities of South Carolina, making it a remnant woodland indicator species.[12][13] S. secundum additionally decreased its occurrence in response to soil disturbance by agriculture in southwest Georgia savannas.[14]
It decreased its frequency in response to soil disturbance by clearcutting and chopping in north Florida flatwoods forests.[15]
This species also decreased its density in response to summer roller chopping in central Florida. It has shown resistance to regrowth in prairie habitat that was disturbed by this activity.[16]
S. secundum was unaffected by summer roller chopping with burning and winter roller chopping in central Florida prairies.[16] It increased its density in response to winter roller chopping with burning in central Florida prairies.[16]
Sorghastrum secundum is frequent and abundant in the Peninsula Xeric Sandhills, Panhandle Xeric Sandhills, North Florida Longleaf Woodlands, and North Florida Subxeric Sandhills community types as described in Carr et al. (2010).[17]
Phenology
Flowering occurs in September and October in Central Florida and northward.[1][5] In southern Florida, blooming begins in July.[5]. Seed germination seems to peak in January and February.[18]
Seed dispersal
This species is thought to be dispersed by gravity. [19]
Seed bank and germination
In south Georgia, S. secundum buried in seed bags had a 26% germination rate after one year, 34% after two, and 9% after four.[18] Following a 1999 cohort in South Carolina, 40% of recruits were alive after three years.[20]
Fire ecology
Fire, especially when followed by freezing or high rainfall, kills or reduces growth in S. secundum. A model suggests longer fire return, such as a three year interval or combination of two, three, and four year intervals will best result in a stable population.[21] Despite this, populations of S. secundum have been known to persist through repeated annual burns.[22][23]
Herbivory and toxicology
S. secundum consists of 2-5% of the diet for terrestrial birds.[24] Fire ants (Solenopsis invicta), do not show an interest in S. secundum seeds.[25] In the southeastern United States, it is also grazed year round.[21] Models suggest herbivory of S. secundum decreases as patched become more isolated from each other[26] and field measurements in South Carolina suggest a given leaf had a 9% probability of herbivory.[27] Lopsided Indiangrass also serves as cover and forage for a number of wildlife species, and it serves a larval host for several species of skipper moth.[28]
Diseases and parasites
Its seeds are more prone to mold destroying it prior to germination compared to other seeds.[18]
Conservation, cultivation, and restoration
Cultural use
Photo Gallery
References and notes
- ↑ 1.0 1.1 1.2 Weakley AS (2015) Flora of the Southern and Mid-Atlantic States. Chapel Hill, NC: University of North Carolina Herbarium.
- ↑ 2.0 2.1 2.2 USDA NRCS (2016) The PLANTS Database (http://plants.usda.gov, 16 January 2018). National Plant Data Team, Greensboro, NC 27401-4901 USA.
- ↑ Plant database: Sorghastrum secundum. (16 January 2018) Lady Bird Johnson Wildflower Center. URL: https://www.wildflower.org/plants/result.php?id_plant=SOSE5
- ↑ Dagley CM, Harrington TB, Edwards MB (2002) Understory restoration in longleaf pine plantations: Overstory effects of competition and needlefall. In: Outcalt KW ed. Proceedings of the eleventh biennial southern silvicultural research conference. General Technical Report SRS-48. Asheville, NC: U.S. Department of Agriculture, Forest Service, Southern Research Station.
- ↑ 5.0 5.1 5.2 5.3 Hall DW (1982) Sorghastrum (Poaceae) in Florida. SIDA 9(4):302-308.
- ↑ 6.0 6.1 Diaz-Toribio, M.H. and F. E. Putz 2021. Underground carbohydrate stores and storage organs in fire-maintained longleaf pine savannas in Florida, USA. American Journal of Botany 108: 432-442.
- ↑ Diaz‐Toribio, M. H. and F. E. Putz. 2021. Underground carbohydrate stores and storage organs in fire‐maintained longleaf pine savannas in Florida, USA. American Journal of Botany 108(3):432-442.
- ↑ Sorrie, B. A. and A. S. Weakley 2001. Coastal Plain valcular plant endemics: Phytogeographic patterns. Castanea 66: 50-82.
- ↑ 9.0 9.1 9.2 Florida State University Herbarium Database. URL: http://herbarium.bio.fsu.edu. Last accessed: June 2021. Collectors: Loran C. Anderson, Wm. G. Atwater, Wilson Baker, E. H. Cooley, A. A. Eaton, R.K. Godfrey, Ann F. Johnson, and R. Kral, O. Lakela, Sidney McDaniel, and Cecil R Slaughter. States and counties: Florida: Collier, Dade, Dixie, Indian River, Liberty, Wakulla, Walton, and Washington. Georgia: Thomas.
- ↑ University of South Florida Herbarium accessed using Southeastern Regional Network of Expertise and Collections (SERNEC) data portal. URL: http://sernecportal.org/portal/collections/index.php Last accessed: June 2021. Collectors: B. F. Hansen, J. Hansen. States and Counties: Florida: Highlands.
- ↑ Carr SC, Robertson KM, Peet RK (2010) A vegetation classification of fire-dependent pinelands of Florida. Castanea 75(2):153-189.
- ↑ Brudvig, L.A. and E.I. Damchen. (2011). Land-use history, historical connectivity, and land management interact to determine longleaf pine woodland understory richness and composition. Ecography 34: 257-266.
- ↑ Brudvig, L.A., E Grman, C.W. Habeck, and J.A. Ledvina. (2013). Strong legacy of agricultural land use on soils and understory plant communities in longleaf pine woodlands. Forest Ecology and Management 310: 944-955.
- ↑ Kirkman, L.K., K.L. Coffey, R.J. Mitchell, and E.B. Moser. Ground Cover Recovery Patterns and Life-History Traits: Implications for Restoration Obstacles and Opportunities in a Species-Rich Savanna. (2004). Journal of Ecology 92(3):409-421.
- ↑ Moore, W.H., B.F. Swindel, and W.S. Terry. (1982). Vegetative Response to Clearcutting and Chopping in a North Florida Flatwoods Forest. Journal of Range Management 35(2):214-218.
- ↑ 16.0 16.1 16.2 Watts, A. C. and G. W. Tanner. 2003. Fire and roller chopping have varying effects on dry prairie plant species (Florida). Ecological Restoration 21(3): 205-207.
- ↑ Carr, S.C., K.M. Robertson, and R.K. Peet. 2010. A vegetation classification of fire-dependent pinelands of Florida. Castanea 75:153-189.
- ↑ 18.0 18.1 18.2 Coffey KL, Kirkman LK (2006) Seed germination strategies of species with restoration potential in a fire maintained pine savanna. Natural Areas Journal 26(3):289-299.
- ↑ Kirkman, L. Katherine. Unpublished database of seed dispersal mode of plants found in Coastal Plain longleaf pine-grasslands of the Jones Ecological Research Center, Georgia.
- ↑ Harrington TB, Dagley CM, Edwards MB (2003) Above- and belowground competition from longleaf pine plantations limits performance of reintroduced herbaceous species. Forest Science 49(5):681-695.
- ↑ 21.0 21.1 Penfield S (2006) Notes on some characteristics of lopsided indiangrass (Sorghastrum secundum). Noss RF (ed) Land of Fire and Water: The Florida Dry Prairie Ecosystem. Proceedings of the Florida Dry Prairie Conference. pp 176-181
- ↑ Robertson, K.M. Unpublished data collected from Pebble Hill Fire Plots, Pebble Hill Plantation, Thomasville, Georgia.
- ↑ Platt, W.J., R. Carter, G. Nelson, W. Baker, S. Hermann, J. Kane, L. Anderson, M. Smith, K. Robertson. 2021. Unpublished species list of Wade Tract old-growth longleaf pine savanna, Thomasville, Georgia.
- ↑ Miller JH, Miller KV (1999) Forest plants of the southeast and their wildlife uses. Southern Weed Science Society.
- ↑ Cumberland MS, Kirkman LK (2013) The effects of the red imported fire ant on seed fate in the longleaf pine ecosystem. Plant Ecology 214:717-724.
- ↑ Brudvig LA, Damschen EI, Haddad NM, Levey DJ, Tewksbury (2015) The influence of habitat fragmentation on multiple plant-animal interactions and plant reproduction. Ecology 96(10):2669-2678.
- ↑ Levey DJ, Caughlin TT, Brudvig LA, Haddad NM, Damschien EI, Tewksbury JJ, Evans DM (2016) Disentangling fragmentation on herbivory in understory plants of longleaf pine savanna. Ecology 97(9):2248-2258.
- ↑ Denhof, Carol. 2011. Understory Plant Spotlight Lopsided Indiangrass Sorghastrum secunda (Elliot) Nash. The Longleaf Leader. Vol. IV. Iss. 1. Page 5